Our Mission
Cells do not live in isolation. Stem cells rely on both local and systemic cues to know when to initiate tissue regeneration. Axons rely on guidance molecules from the target cells to find their path. Cells of multiple lineages often coordinate proliferation to expand overall organ size during development.
Our goal is to discover the principles and molecular nature of cell-cell interactions governing development, regeneration, and injury repair. We use the mammalian skin as our model, a highly accessible organ with diverse cell types and multiple populations of somatic stem cells.
Research
Our Model
Mammalian skin serves as a physical barrier protecting organisms from injury, infection, and dehydration. The skin also regulates body temperature and receives complex sensory inputs. These diverse functions are made possible by a rich array of cell types. The epidermis, the hair follicle, and the melanocyte lineage contain tissue-resident stem cells and are among some of the most highly regenerative tissues in adult mammals. These stem cells regenerate in a rich environment filled with fibroblasts, immune cells, neurons, blood vessels, muscle, and adipocytes. The cell-cell interactions that occur in this organ are complex and vast in number. Therefore, the mammalian skin provides us with a plethora of opportunities to explore how cells from diverse lineages coordinate their behaviors to build and maintain a functional organ.

Our Approach
We combine numerous experimental approaches, including high-resolution imaging, lineage-tracing, mouse genetics, in utero surgery, flow cytometry, in vivo gene-editing, single-cell or pooled RNA-seq, and cell culture.
Projects
The Cellular and Molecular Identity of the Stem Cell Niche
Stem cell behaviors are heavily influenced by the niche microenvironment, where stem cells reside. However, the identity of niche factors and niche cell types remains elusive for many mammalian stem cells. We have established tools and strategies to manipulate gene expression in numerous skin cell types and are using them to investigate reciprocal interactions between stem cells and niches. This research will define mechanisms that could be targeted to promote tissue regeneration or wound healing. Our current research focus in this area includes:
Feedback regulation by stem cell progeny
We have pioneered studies to identify stem cell progeny as important regulators of activity of parental stem cells (Hsu et al., Cell 2011, Hsu et al., Cell 2014). Currently, we are identifying specific signaling factors that govern this feedback regulation.
Identification of novel niche cell types and signals
With new tools that we established, we are systematically identifying novel cell types and secreted factors that govern stem cell quiescence, promote stem cell self-renewal, and instruct stem cell fate decisions.
Modification of stem cell behavior to enhance wound repair
More than 100 million people develop scars each year as a result of trauma, surgery, or burns. At least 6 million people suffer from chronic non-healing wounds (including diabetic foot ulcers and bedsores). We are applying what we have learned about how the niche regulates skin stem cells to develop novel wound healing strategies.

Function and Biology of Transit-Amplifying Cells
Somatic stem cells are often quiescent but occasionally divide to generate “transit-amplifying cells” (TACs). TACs then undergo rapid division and expansion to produce a large quantity of differentiated cells downstream.
Since TACs are the workforce of tissue production while stem cells are mostly dormant, TACs are located at an ideal juncture to orchestrate production of their own downstream progeny with critical changes in surrounding niche cell types that accommodate and support a regenerating tissue (see our review comparing TACs in the blood and the skin (Zhang and Hsu WIREs Developmental Biology). Current projects relevant to TAC biology include:
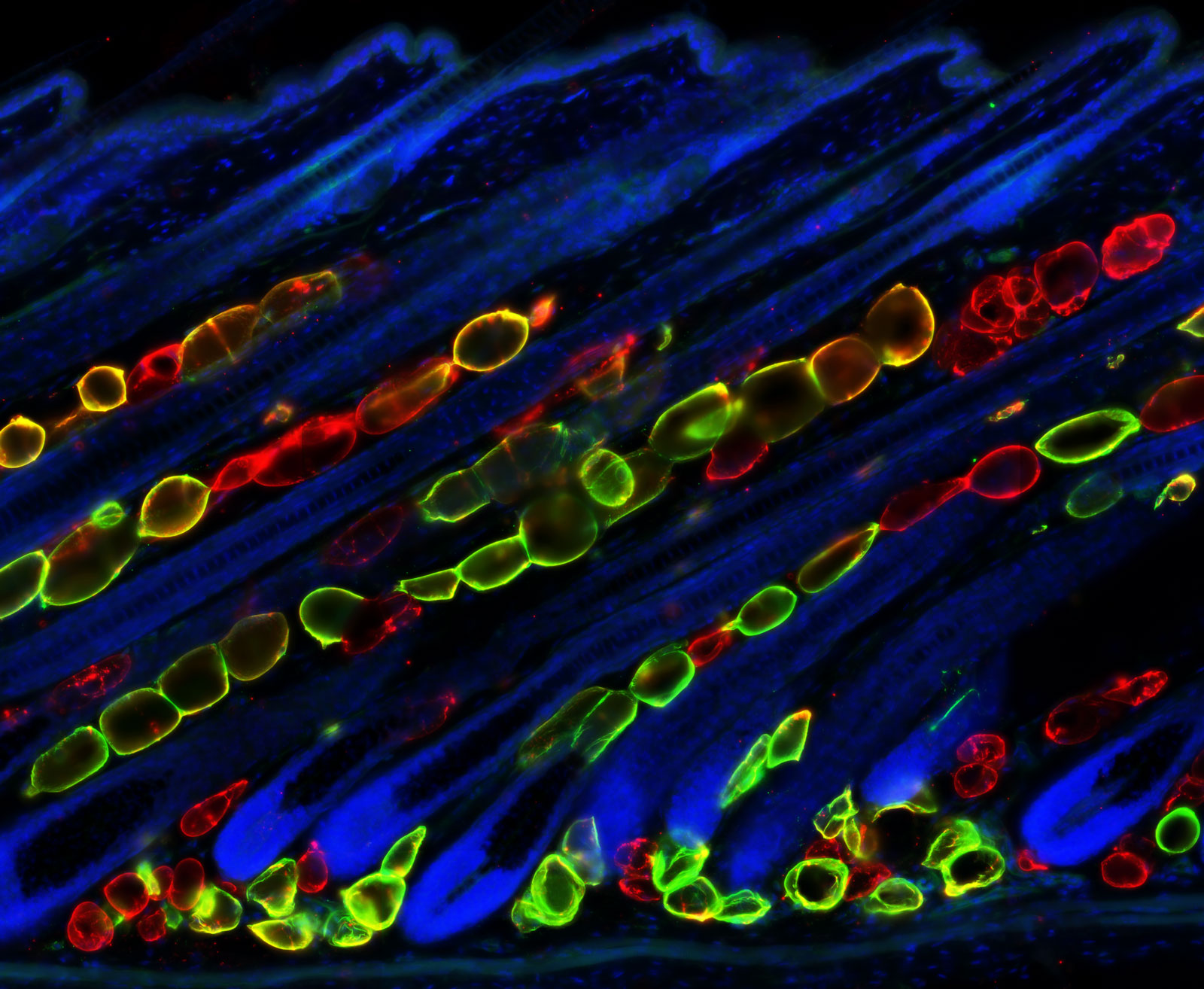
TACs of one tissue control changes in many neighboring tissues
How tissues of different lineage grow together in an organ is a fundamental question in developmental biology. We discovered that hair follicle TACs control growth of the hair follicle itself and production of neighboring dermal adipocytes, coupling activity of two lineages (Zhang et al, G&D 2016). Currently, we are defining TAC function in regulating other tissues in the skin.
TACs and chemotherapy-related side effects
The highly proliferative nature of TACs is what makes them, not slow-cycling stem cells, a primary target of cytotoxic chemotherapies. Thus, understanding TAC function may inform strategies to prevent chemotherapy-related damage to tissues and organs. By studying the biology of TACs, we have discovered the root causes for several chemotherapy-related side effects, including hair loss, delayed wound healing, and susceptibility to infections. We are working on the cellular and molecular mechanisms leading to these phenotypes.
How Systemic Changes Regulate Stem Cell Behavior
Stem cells must respond not just to the niche but to systemic signals in response to varying demands (for example, stress, pregnancy, or aging). Our lab has established approaches to determine if and how changes in systemic factors influence cell-cell interactions, stem cell behavior, and repair of injury in the skin. We have defined several circulating factors crucial for skin stem cell function and are now determining whether these factors act directly or through the niche to regulate stem cell activity and tissue regeneration.
funding & awards













